Climate change is one of the most urgent global issues of our time and has significant implications for human health and the healthcare system. The World Health Organization (WHO) describes climate change as “the greatest threat to humanity,”[1] highlighting the close connection between climate and health. The increase in extreme weather events, such as heatwaves, storms, and floods, can directly and indirectly cause severe health problems, including heatstroke, respiratory diseases, infections, and mental stress. This leads to a heightened demand for medical care requiring targeted preparation and adaptation in healthcare systems.
At the same time, the healthcare system is confronted with determining and reducing its own environmental impact. Accounting for 5.2% of nationwide greenhouse gas emissions, the healthcare sector in Germany emits more greenhouse gases than the air and maritime industries combined[2]. Greenhouse gases are significant contributors to climate change, and reducing emissions is a major lever to halt further global warming.
However, achieving direct emission reductions in healthcare poses a unique challenge as cost-saving measures must not negatively impact the quality of patient care. An example illustrating how climate protection and healthcare goals can be aligned, is seen in the treatment of respiratory conditions such as bronchitis, asthma, or COPD. In these cases, inhalation devices are usually prescribed to alleviate or cure the symptoms. Many people are unaware that certain inhalation therapy options can contribute substantially to their individual greenhouse gas footprints. For instance, the use of a pressurized metered dose inhaler (pMDI) for inhalation therapy emits more CO2 in a year than a short-haul flight (see figure 1). Potentially more environmentally friendly are dry powder inhalers (DPI), as their annual use generates 20-30 times less CO2 as the use of pMDIs[3]. Unfortunately, DPIs cannot completely replace the use of pMDIs, as not all patients can fulfill the specific handling and physical requirements for inhalation.
In the consideration of the right therapy, the well-being of the environment and that of patients must not be pitted against each other. To offer inhalative preparations in both an environmentally friendly pressurized metered dose inhaler and as a dry powder inhaler, our partner, the Chiesi Group, a research-oriented and family-owned pharmaceutical company, decided in 2019 to invest 350 million Euros. This investment aims to develop a pressurized gas inhaler (DA) with low greenhouse gas emissions and to replace the current propellant (HFA 134a) with a new, more climate-friendly propellant (HFA-152a) by the end of 2025. Studies have already demonstrated that this new formulation exhibits comparable safety and tolerability to the previously used propellant. The deployment of the new pMDI generation could reduce the CO2 footprint of these inhalers by up to 90 %, marking a step towards a more sustainable healthcare system[4].
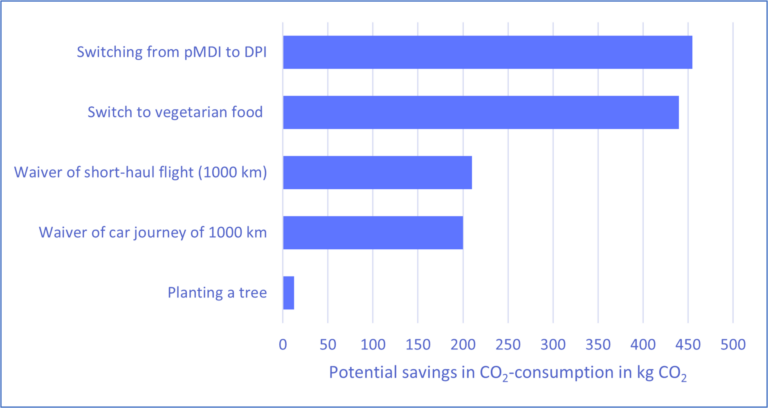
Figure1: Various options for CO2 savings[5]
High temperatures and changing weather conditions pose an additional burden for individuals with respiratory or lung diseases[6], and high air pollution is linked to increased mortality[7]. Chiesi Group is strategically investing to develop the necessary innovations to achieve environmental goals while meeting patient needs. A stable and well-treated patient requires fewer resources in the long run, embodying the holistic approach integral to the corporate philosophy of the Chiesi Group. The company has also registered as a Benefit Corporation in countries where possible and, as one of the few global pharmaceutical companies, obtained B CorpTM certification in 2019.
Addressing climate change is not a competition, and all stakeholders in the industry are encouraged to collectively take further steps towards a carbon-neutral and sustainable healthcare system. We take pride in having such partners in our network and welcome a bold and solution-oriented discussion on the topic.
Feel free to contact us if you are interested.
[1] https://www.who.int/news-room/fact-sheets/detail/climate-change-and-health
[2] https://noharm-global.org/documents/health-care-climate-footprint-report
[3] https://www.prescqipp.info/umbraco/surface/authorisedmediasurface/index?url=%2fmedia%2f5721%2f295i-inhaler-carbon-footprint-22.pdf
[4] https://www.chiesi.de/en/erstes-dosieraerosol-mit-reduziertem-treibhauseffekt-ist-auf-kurs-zum-wohle-der-patient*innen-und-der-umwelt-
[5] Klimabewusste Verordnung von inhalativen Arzneimitteln, DEGAM S1-Handlungsempfehlung, AWMF-Register-Nr. 053-059
[6] Climate change and respiratory health: a European Respiratory Society position statement
Ana Maria Vicedo-Cabrera et al., European Respiratory Journal Aug 2023, 62 (2) 2201960; DOI: 10.1183/13993003.01960-2022
[7] https://www.eea.europa.eu/publications/environmental-burden-of-cancer/air-pollution